Cosmetics Compliance
Done Right
Compliance in US, Canada, EU, and UK is complex and constantly changing. Our regulatory experts and services can help you get and stay compliant.
Evolving Regulations Create Risk
Cosmetic regulatory requirements are constantly evolving and unique to each region. Falling behind can delay launches, block business expansion, or create expensive regulatory actions. Here are just a few examples of key requirements that companies must not overlook:
- US FDA’s new MoCRA regulation mandates registration, labeling, product listings, and adverse event reporting.
- California requires ingredient-level disclosures for Prop 65.
- EU, UK, and Canada require a local Responsible Person, safety assessments, labeling controls, and bilingual packaging.
Launch in the US with Confidence
The new US FDA and California-specific regulations add complexity to your business. With Registrar as your compliance partner, you can:
- Enter the US market faster with complete FDA MoCRA compliance.
- Avoid relabeling and delays with accurate formula and label reviews.
- Reduce risk with GMP-aligned documentation and processes.
- Reduce enforcement risk with California-specific label and claims review.
- Stay aligned nationwide by harmonizing MoCRA + California requirements.

The FDA’s Modernization of Cosmetics Regulation Act (MoCRA) created many new requirements. We can help you gain and maintain compliance including facility registration, product listings, adverse event reporting, label compliance, formulation & GMPs, and compliance training.

California is the largest US cosmetics market and has strict regulations beyond the FDA. We can help you meet California regulations including formula reviews, ingredient & cumulative concentration analysis, and Prop 65 warning label requirements.
Expand into Europe and UK with Confidence

The EU and UK set the global standard for cosmetic safety. We make compliance effortless so you can focus on growth. With Registrar as your partner, you can:
- Access EU/UK markets quickly with complete, compliant CPSRs.
- Gain legal protection and be audit ready with your own Responsible Person (RP).
- Pass retailer checks easily with audit-ready PIFs.
- Eliminate rework with accurate labeling and claims guidance.
- Remove administrative burden through expert-managed CPNP/SCPN filings.
Enter Canada with a Trusted Compliance Partner
Canada demands bilingual labeling and strict ingredient compliance. We ensure your products meet the mark. With Registrar as your partner, you can:
- Avoid approval delays with timely CNF filings.
- Protect your launch timeline with Hotlist ingredient checks.
- Reduce relabeling costs with fully compliant English/French labels.
- Meet local requirements through a Canadian Responsible Person.

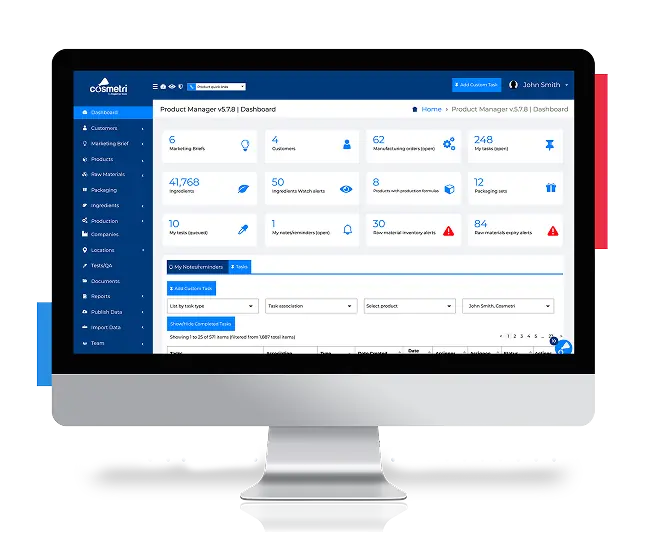
Build Compliant Products with Cosmetri® Software
Registrar’s Cosmetri® product lifecycle management software is your centralized system for formulation, documentation, and compliance.
- Shorten internal review cycles.
- Reduce formulation risks.
- Keep documentation organized and compliant.
A Single Platform for Global Adverse Event Management
Our Adverse Event Management (AEM) solution enables the capture, evaluation, and reporting of cosmetic adverse events so you stay compliant in every market.
With Registrar’s AEM software, you can:
- Centralize adverse event intake from all channels.
- Assess reportability using expert medical review.
- Maintain defensible records for audits and inspections.
Professional Training for Your Compliance Q&A Staff
Our expert-led online training in GMPs, ISO 9001, ISO22716, GLPs, and FDA MoCRA regulations ensure your staff have the knowledge and expertise they need.

Schedule a Free Consultation
Ready to take your beauty products across borders? Submit this form to discover how Registrar can help.
